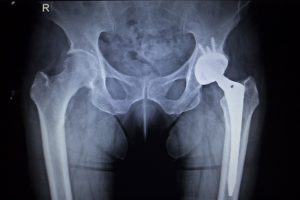
In recent months, the OMNI Apex K2 Hip Replacement implants have been under investigation as deterioration and premature failure of these medical devices is discovered. Any recipients of these modular hip replacement parts could be at risk in as little as one year after implant surgery.
What is the OMNI Apex K2 Hip Replacement System?
The OMNI Corporation, formerly known as OMNIlifescience, Inc. initially released the Apex K2 Modular Hip Replacement System for sale to patients in 2004, when it was first cleared by the US Food and Drug Administration (FDA).
The FDA approved the modular system for use in patients suffering from fractures, osteoarthritis, congenital deformity, and rheumatoid arthritis. Patients undergo surgery to have the devices implanted, replacing original joints. Because it is a modular system, it is made up of different combinations of possible head, neck and stem pieces, customized to fit each patient’s body. OMNI advertises their Apex K2 modular hip system as offering up to 96 different variations of the head, neck and stem.
The pieces of the OMNI Apex K2 Hip system are metal and ceramic. The stem is the bottom piece of the unit, made of titanium, and is fitted into the thigh bone. The top is known as the head or the ball, which is either metal or ceramic. This head piece is connected via a small metal collar piece called the neck. When fitted together, the head replaces the rounded part of the femur bone, and fits inside the hip socket to rotate, giving the patient range of motion.
KEEFE LAW FIRM’S VERDICTS & SETTLEMENTS
Device Deterioration Causing Metal to Enter Bloodstream
The biggest point of weakness in the OMNI Apex K2 Hip Replacement System is the wearing down of the metal pieces against one another. When the metal stem moves against the neck and head pieces, there has been some deterioration of the metal surface. While the modular design of the Apex K2 system allows better fit, the separate pieces cause friction, which leads to this deterioration.
As the metals rub against each other, small particles can come dislodged. These metal particles can damage surrounding tissue and can even enter the blood stream, resulting in a type of metal poisoning called Metallosis. Patients can experience pain, swelling, hearing loss, heart failure, vision weakness, thyroid problems and nerve issues when suffering from Metallosis.
Once diagnosed with Metallosis, patients need to undergo a second revision surgery to replace the metal parts of the implant with plastic or ceramic materials. However, some patients still suffer from loss of mobility and enduring pain even after revision surgery.
Modular Pieces More Likely to Break
Even in cases where patients do not suffer from Metallosis, they still may need to undergo revision or subsequent replacement surgeries to fix broken pieces of the Apex K2 Modular Hip System. The stem piece seems to be most vulnerable to breakage in two points. Where the stem fits into the neck, there have been reports of stem breakage, resulting in emergency surgery. Also, the stem pushing against the femur has cause fractures of the femur around the stem piece.
Both types of breakage force the patient into an emergency surgical situation that could result in further joint damage.
No Recall, Yet Pulled from Australian Market
When similar problems of deterioration and breakage occurred with the Stryker Rejuvenate and ABG II hip devices, Stryker recalled their components. However, OMNI has not yet made any move to recall their similarly problematic devices from the market.
While these devices are still being used in the US, they have been pulled from the Australian Market. The Australian Register of Therapeutic Goods (ARTG) performs the same services as the United States’ FDA. The ARTG cited “a higher than expected revision rate” as the reason from pulling the device for Australian patients in 2012. Instead of giving patients lasting relief from hip pain, studies found that the majority of the Apex K2 modular hip pieces showed failure in one to three years.
Recourse if You Have a Failed OMNI Apex K2 Hip Replacement
Many patients who are suffering premature failure of hip replacement devices have filed successful lawsuits against the device manufacturers. Whether the failure is in only the device itself and needs to be replaced too soon, or if the deterioration of the metal on metal components has caused other health factors, patients can file lawsuits to recoup medical costs and compensation for suffering.
There are many factors to consider if you have a failed OMNI Apex K2 modular hip device. Contact our law offices today to review your case and decide on a course of action.