Implant and Medical Device Lawsuits
Experienced Medical Device Injury Lawyers
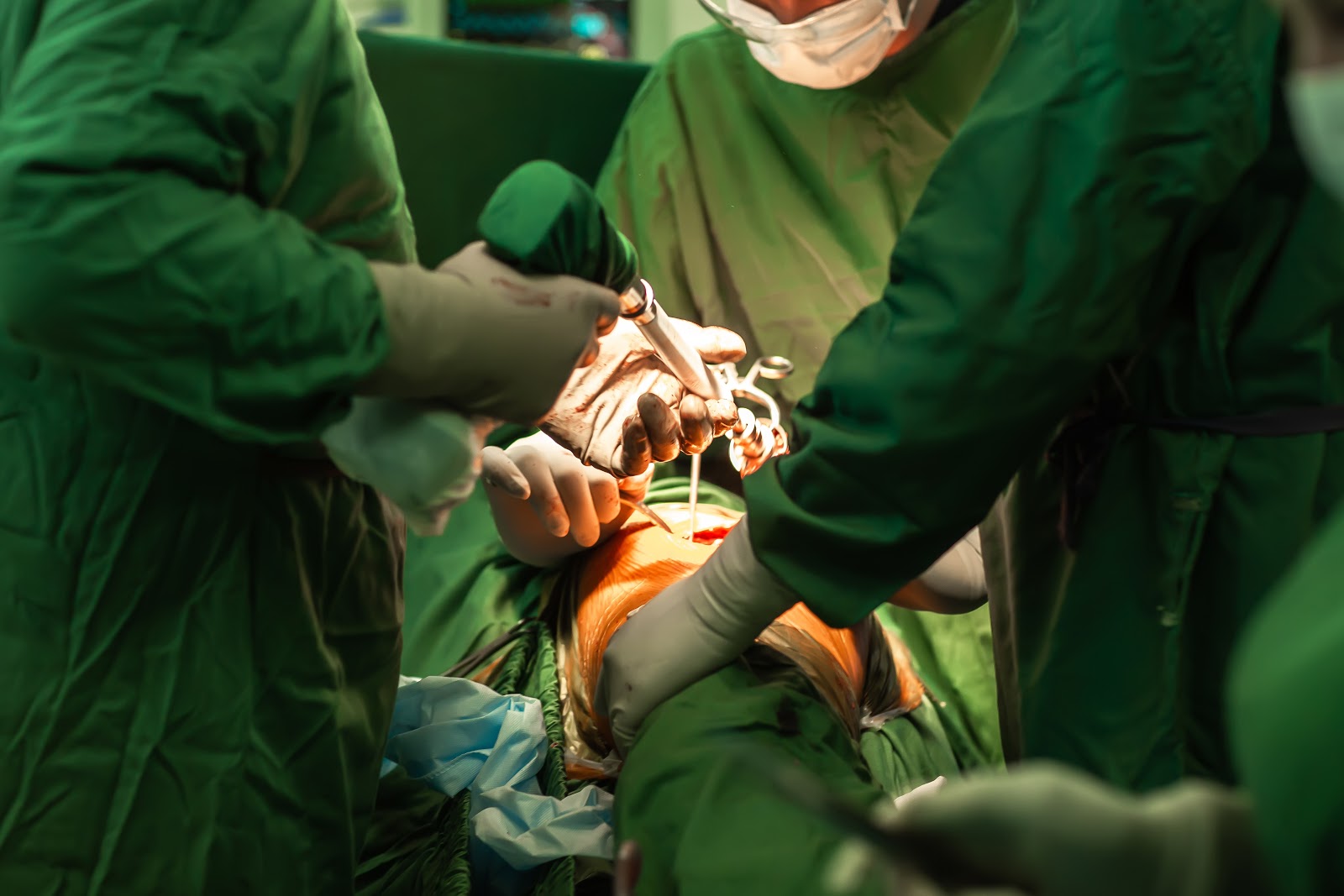 A medical device is an implant, instrument, apparatus, machine or tool meant to diagnose, prevent, mitigate, treat or cure a disease, illness, injury or other health problem. Today, there are tens of thousands of medical devices on the market, ranging from thermometers to neurostimulators. For the most part, medical devices and implants improve patient health and quality of life for millions of people around the world.
A medical device is an implant, instrument, apparatus, machine or tool meant to diagnose, prevent, mitigate, treat or cure a disease, illness, injury or other health problem. Today, there are tens of thousands of medical devices on the market, ranging from thermometers to neurostimulators. For the most part, medical devices and implants improve patient health and quality of life for millions of people around the world.
However, a perfect storm of corporate negligence and greed combined with a flawed U.S.Food and Drug Administration (FDA) approval process has enabled hundreds of types of defective devices to be implanted in countless people, resulting in pain, injury and illness that often cannot be reversed.
The Most Harmful Medical Devices
The medical device industry is one of the most lucrative and expected to reach nearly $600 billion globally by 2024. The incentive for medical device companies like Smith & Nephew, Stryker, J&J and Zimmer to quickly get products to market is huge. In this rush to manufacture, market and sell medical devices some companies take shortcuts, knowing that the FDA will not require them to obtain pre-market approval. Due to this lack of clinical trials and testing on humans, there are hundreds of implants and devices that have caused significant harm and even death to patients. Among the most egregious are:
Hip Replacements
- Smith & Nephew Birmingham Metal-on Metal Hip Resurfacing System
- Depuy Pinnacle Ultamet Metal on Metal Hip Implant
- Stryker LFIT V40 Femoral Head
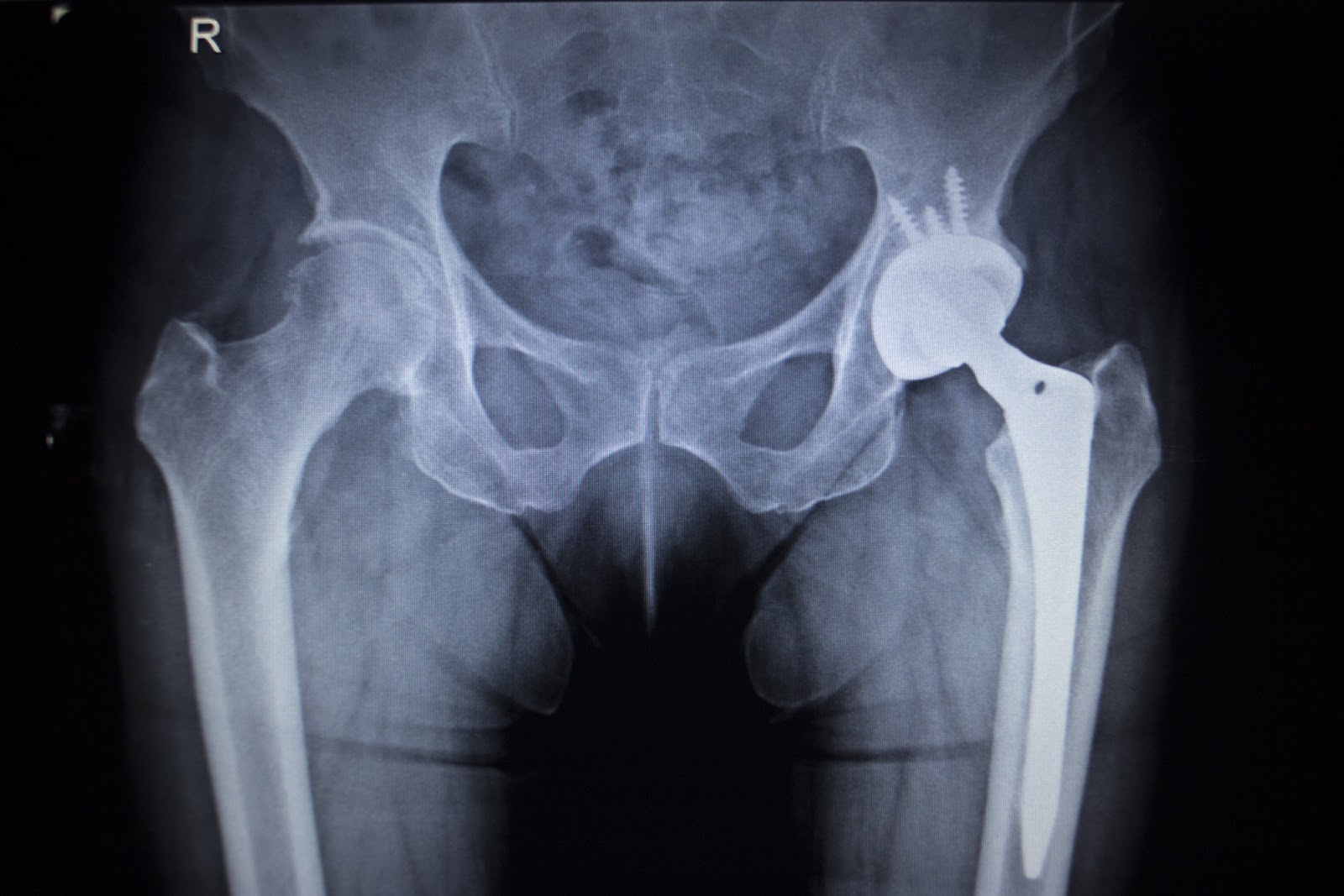 Metal-on-metal hip implants have been found to have a higher premature failure rate than other types of implants. The only way to correct this is to have costly and painful revision surgery where the entire hip replacement system is removed and a new one is implanted. Often the bone loss, necrosis, diminished mobility and other illnesses and injuries caused by the metal on metal implant cannot be reversed. Metal-on-metal implants are also known to release chromium and cobalt ions into the patient’s bloodstream. These dangerous substances can cause heart disease, neurological impairment, cognitive impairment and other serious health issues.
Metal-on-metal hip implants have been found to have a higher premature failure rate than other types of implants. The only way to correct this is to have costly and painful revision surgery where the entire hip replacement system is removed and a new one is implanted. Often the bone loss, necrosis, diminished mobility and other illnesses and injuries caused by the metal on metal implant cannot be reversed. Metal-on-metal implants are also known to release chromium and cobalt ions into the patient’s bloodstream. These dangerous substances can cause heart disease, neurological impairment, cognitive impairment and other serious health issues.
More than $1.5 billion has been set aside or paid out to victims hurt by the Pinnacle hip replacement; Smith & Nephew is facing 587 product liability lawsuits alleging their Birmingham Hip Resurfacing System fails prematurely, particularly affecting women, and also causes metallosis and pseudotumors.
In 2016, Stryker Corp. recalled its LFIT v40 Femoral Head (the part of a hip replacement that attaches to the thigh bone) after 635 plaintiffs filed claims alleging early failure, necessitating revision surgery. These cases were subsequently consolidated in a multidistrict litigation in Massachusetts and a settlement agreement was reached in November 2018. The initial settlement terms are confidential and will not be publicly disclosed. Settlement values are believed to vary depending on certain case specific criteria including the degree of harm and the extent of permanent injuries that were suffered.
Knee Replacements
- DePuy Attune Knee Replacement System
- B. Braun Aesculap Knee Replacement
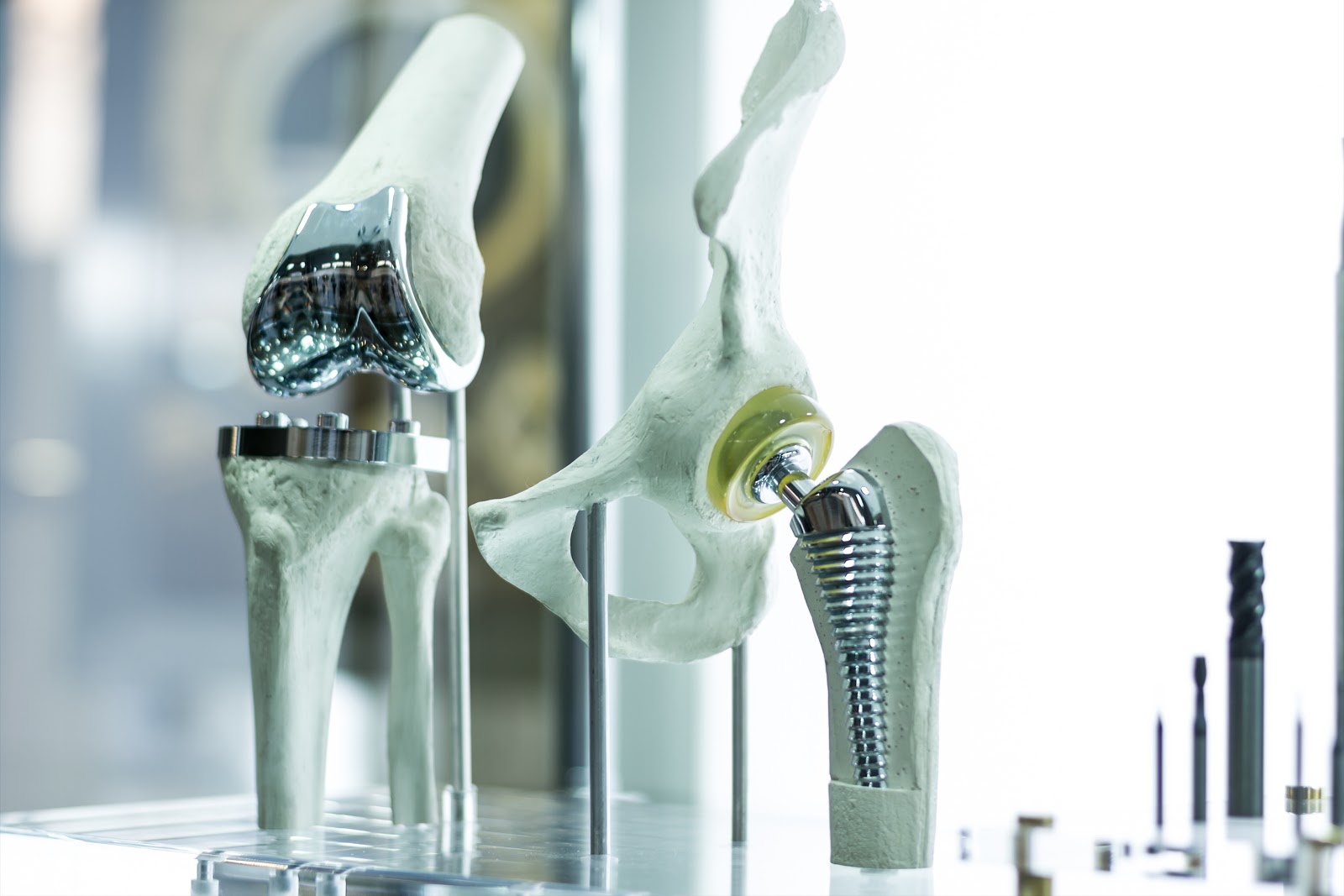 Documented premature failure of DePuy’s Attune Knee Replacement System caused the FDA to recall it in June 2015. The Attune Knee Replacement had a product defect in which the device “de-bonds” or simply put, becomes unglued, causing the device to loosen and the entire system to destabilize. The only option for the patient is revision surgery. Numerous lawsuits have been filed and are expected to commence in 2019.
Documented premature failure of DePuy’s Attune Knee Replacement System caused the FDA to recall it in June 2015. The Attune Knee Replacement had a product defect in which the device “de-bonds” or simply put, becomes unglued, causing the device to loosen and the entire system to destabilize. The only option for the patient is revision surgery. Numerous lawsuits have been filed and are expected to commence in 2019.
B. Braun is the most recent addition to the list of defective medical device manufacturers. So far, 30 plaintiffs have filed suit alleging premature failure of the device and failure to warn patients that the Aesculap knee can become loose due to failure of the system to bond.
Pelvic or Transvaginal Mesh
- Boston Scientific
- Coloplast
- CR Bard
 In 2002, the FDA cleared pelvic mesh for use in treating pelvic organ prolapse (POP) a fairly common medical problem among women. Pelvic mesh is made from polypropylene and inserted into the vaginal wall. It was categorized by the FDA as a Class II moderate-risk device and did not undergo clinical testing in humans. Soon after its introduction, reports emerged of considerable pain, critical injuries and death among women who had been implanted with pelvic mesh.
In 2002, the FDA cleared pelvic mesh for use in treating pelvic organ prolapse (POP) a fairly common medical problem among women. Pelvic mesh is made from polypropylene and inserted into the vaginal wall. It was categorized by the FDA as a Class II moderate-risk device and did not undergo clinical testing in humans. Soon after its introduction, reports emerged of considerable pain, critical injuries and death among women who had been implanted with pelvic mesh.
According plaintiffs’ claims filed against manufacturers such as Johnson & Johnson, Boston Scientific and CR Bard, pelvic mesh can degrade, come loose and push through a woman’s skin and tissue, causing bleeding, infection, painful intercourse, incontinence and other devastating injuries and illnesses. Finally, on April 12, 2019 after much public outcry, the FDA stopped the sale and use of pelvic mesh in the U.S. Litigation over pelvic mesh is one of the biggest mass tort cases in U.S. history; seven medical device manufacturers, including Boston Scientific and Johnson & Johnson are paying nearly $8 billion to resolve the claims of more than 100,000 women. Product liability lawsuits continue to be filed–a second wave of litigation against Boston Scientific relating to their Obtryx sling alleging that they intentionally sold a defective medical device, failed to warn women and physicians about its safety risks and intentionally concealed information.
Hernia Mesh
- C.R. Bard
- Cook Medical
- Ethicon Physio Mesh
 Hernias are typically caused by a combination of pressure and an opening or weakness of muscle or connective tissue. This pressure can push an organ or tissue through an opening or a weak spot in a person’s stomach, groin, diaphragm or other area of their body. Today, some one million hernia repair procedures are performed in the U.S. each year. Hernia mesh is now used in 80% of all hernia repair surgery and has been employed by physicians for over 100 years to reinforce weakened or damaged tissue. Manufacturers of hernia mesh include Bard Davol, Inc., Ethicon, Atrium, B.Braun and Covidien.
Hernias are typically caused by a combination of pressure and an opening or weakness of muscle or connective tissue. This pressure can push an organ or tissue through an opening or a weak spot in a person’s stomach, groin, diaphragm or other area of their body. Today, some one million hernia repair procedures are performed in the U.S. each year. Hernia mesh is now used in 80% of all hernia repair surgery and has been employed by physicians for over 100 years to reinforce weakened or damaged tissue. Manufacturers of hernia mesh include Bard Davol, Inc., Ethicon, Atrium, B.Braun and Covidien.
While hernia mesh reduces the rate of hernia recurrence. However, due to incompatibility with the human body caused by the chemical and structural nature of the mesh, many patients experienced serious adverse effects such as pain, infection, perforation, adhesion to internal organs, and bowel obstruction, prompting the filing of thousands of adverse event reports with the FDA.
Injured By A Defective Medical Device? Contact Our Attorneys Today
At Keefe Law Firm, our product liability attorneys have extensive experience litigating cases involving defective medical devices and implants. In one case, our lawyers obtained a settlement of over $5 million for victims of defective DePuy hip implants. If you or someone you love suffered injuries as a result of product defects in a medical device, our law firm can help. We offer free consultations over the phone or at our offices in Red Bank, New Brunswick and Newark. We have also helped clients obtain multi-million dollar settlements for defective medical devices.
